 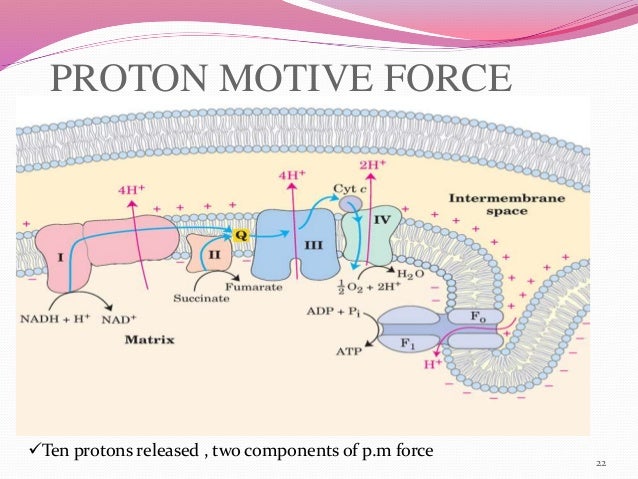 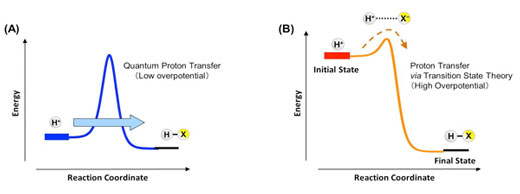
De La Salle University Hideaki Kasai Osaka University Abstract We explore the initial driving. A free energy correlation supports this conclusion. Initial driving force for proton transfer in Nafion Authors: Muneyuki Tsuda Nelson B. These are ascribed to a strong coupling of the electron transfer with the concomitant, thermodynamically favorable proton transfer. Comparisons with rates calculated with the use of Marcus theory reveal that reactions of +, where the electron transfer product is the highly acidic 2+, show rate enhancements of several orders of magnitude. Rate-Driving Force Relationships in the Multisite Proton-Coupled Electron Transfer Activation of Ketones J Am Chem Soc. A free energy correlation supports this conclusion.ĪB - The rates of reduction of the nickel(III) oxime-imine complexes, + and 2+ by the corresponding iron(II) species are determined. In addition to increasing the driving force for proton transfer, enhancing the basicity of the carboxylate also decreases the equilibrium proton donor-acceptor. Here we present a detailed kinetic study of the multisite proton-coupled electron transfer (MS-PCET) activations of aryl ketones using a variety of Brønsted acids and excited-state Ir. Comparisons with rates calculated with the use of Marcus theory reveal that reactions of +, where the electron transfer product is the highly acidic 2+, show rate enhancements of several orders of magnitude. The rate dependence on the driving force for both electron transfer and proton transfer-employing different combinations of oxidants and bases-was used to establish experimentally the concerted. N2 - The rates of reduction of the nickel(III) oxime-imine complexes, + and 2+ by the corresponding iron(II) species are determined. Understanding proton transfer in nanochannels on the molecular level remains a fundamental issue in various areas of science and technology, from biological systems to electrochemical energy conversion 1, 2, 3. In each catalytic cycle, four pumped protons are transferred from the N-side to the P-side of the membrane. 
The authors thank CAPES (Brazil) for a maintenance grant to M.C.M.L. Different redox states of the metal centers formed by electron transfer provide the driving force for proton pumping. electrodes reveal unusual reaction rate / driving force effects that are. T1 - Driving force effects in proton coupled electron transfer EVALUATING INTERFACIAL CHARGE TRANSFER KINETICS AND ENERGETICS VIA LASER BASED. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
